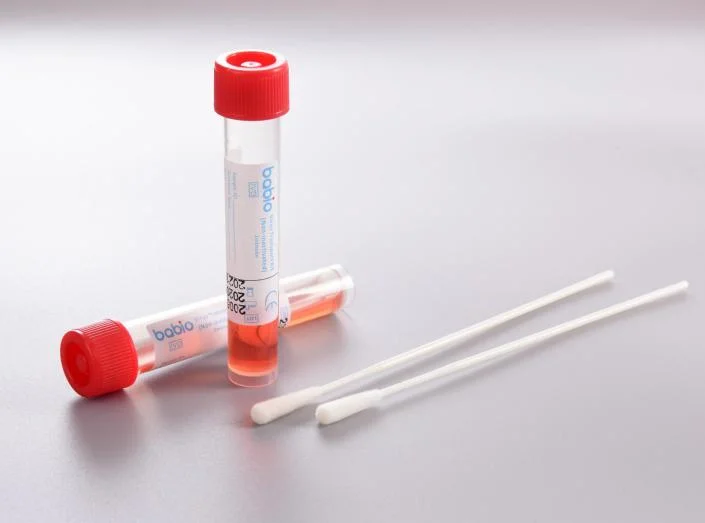
1.Virus Transport Kit (Non-inactivated)
1. ROOM TEMPERATURE STABLE
2.UNIQUE MEDIA FORMULATION
Improved formulation of hanks solution,combined with multiple antibiotics to inhibit the reproduction of bacteria and fungal flora
3.SAFE AND RELIABLE SWABs UNIQUE BREAKPOINT DESIGN
4.SAFE, SHATTERPROOF, STAND UP tubes
Thickened design, with distinctive internal conical shape enabling centrifugation of samples. No DNase, RNase and toxic residue.
5.Multiple specifications
Large media fill volume allows for multiple tests on the same specimen.small volume prevents sample dilution.
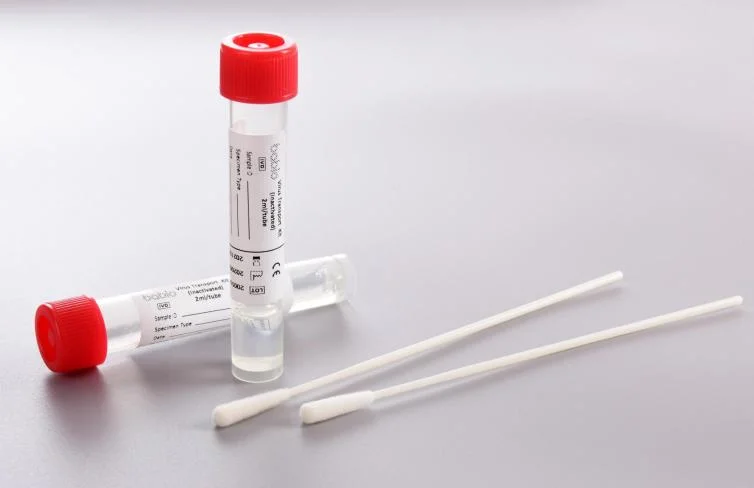
2.Virus Transport Kit (Inactivated)
1.SAFETY
Contains highly efficient lysate for rapid lysis and inactivation of viruses, Eliminate biosafety hazards.
Synchronous sampling and deactivation
3.ROOM TEMPERATURE STABLE
Store at room temperature without refrigeration
4.STABILIZED NUCLEIC ACID
Unique media-stable formulation, Long-term stable storage of nucleic acids
Large media fill volume allows for multiple tests on the same specimen.small volume prevents sample dilution.
| babio Virus Transport Kit | |||
| Type | Non-inactivated | Inactivated | |
| Specifucation | 1ml/tube,2ml/tube,3ml/tube,3.5ml/tube,5ml/tube,6ml/tube | 1ml/tube,2ml/tube,3ml/tube,3.5ml/tube,5ml/tube,6ml/tube | |
| Package | Pkg of 20,Pkg of 50,Pkg of 100,Pkg of 200,Pkg of 300,Pkg of 500 | Pkg of 20,Pkg of 50,Pkg of 100,Pkg of 200,Pkg of 300,Pkg of 500 |
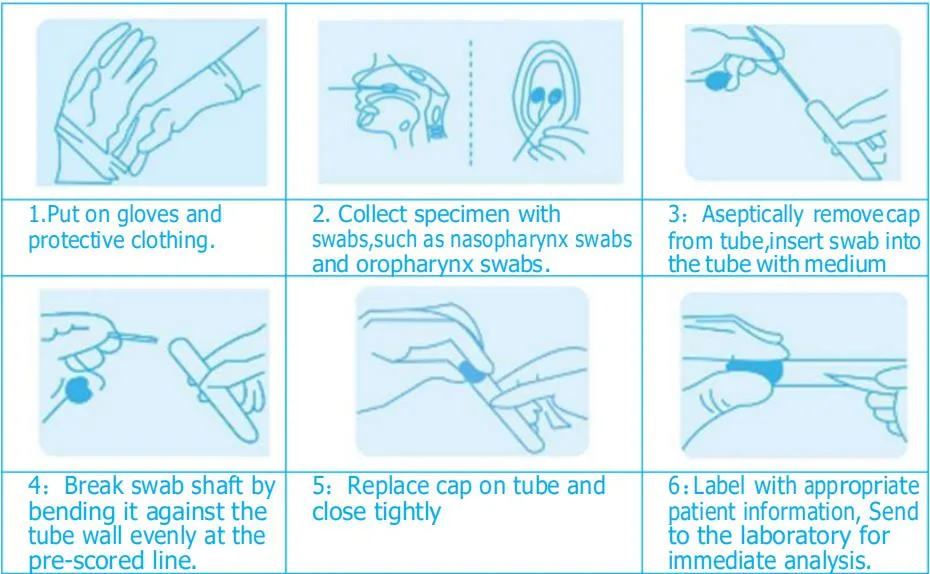


Jinan Babio Biotechnology Co., Ltd.
Founded in 2003, Jinan Babio Biotechnology Co., Ltd. is a professional high-tech enterprise which concentrates on the R&D, manufacturing and selling of in vitro diagnostic reagent. The company was successfully listed on the NEEQ (National Equities Exchange and Quotations) in 2014 (security name: Baibo Biotechnology, stock code: 830774). The company has established a strict quality management system and passed ISO9001:2008 and ISO13485:2003 international quality system certifications. The company has internationally advanced fully automatic production equipment and standard purification workshop of pharmaceutical grade. Baibo has strong biotechnology power, and currently has more than 72 independent intellectual property rights, it undertakes a number of national scientific research projects and has 91 medical device registration certificates approved by CFDA. The company's products mainly involve virus sampling, rapid detection kits, microbial detection reagents, POCT detection reagents, biochemical diagnostic reagents, diagnostic equipment and so on. The pandemic out broke all over the world in 2020, Babio has been actively involved in the R&D and manufacturing of sampling and testing reagents. Currently, the company's products of virus transport media, nasal/oral sampling swabs, and the products' quality has been recognized by the medical institutions and laboratories of various countries, In the future, Babio will be committed to providing better technology and services, and continue to struggle for the human health.